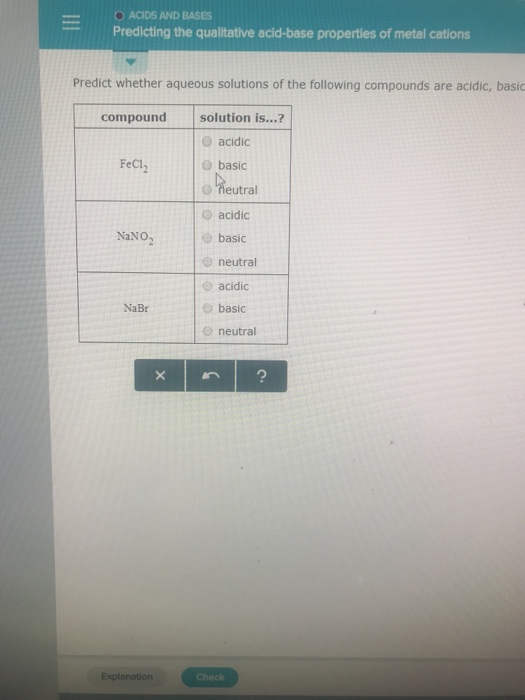
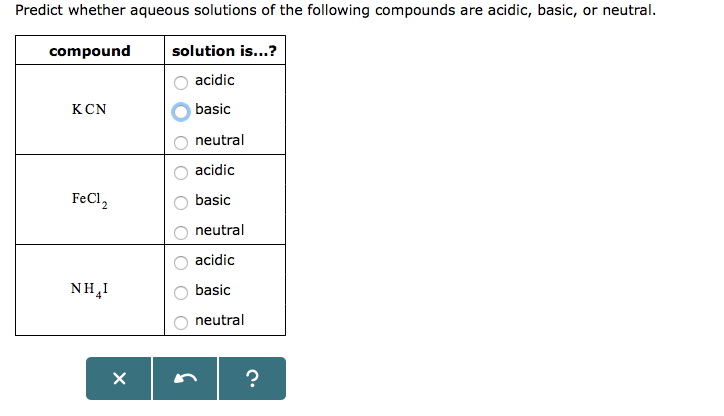
SOLVED: Classify each chemical reaction: 1|c| reaction 2c| type of reaction (check all that apply) FeSO4(a q)+PbCl2(a q) →FeCl2(a q)+PbSO4(s) □ combination □ precipitation □ single replacement □ combustion □ double replacement

Acids and Bases Chapter 14 Acids and Bases. Acids and Bases Some Definitions Arrhenius Acid:Substance that, when dissolved in water, increases the concentration. - ppt download
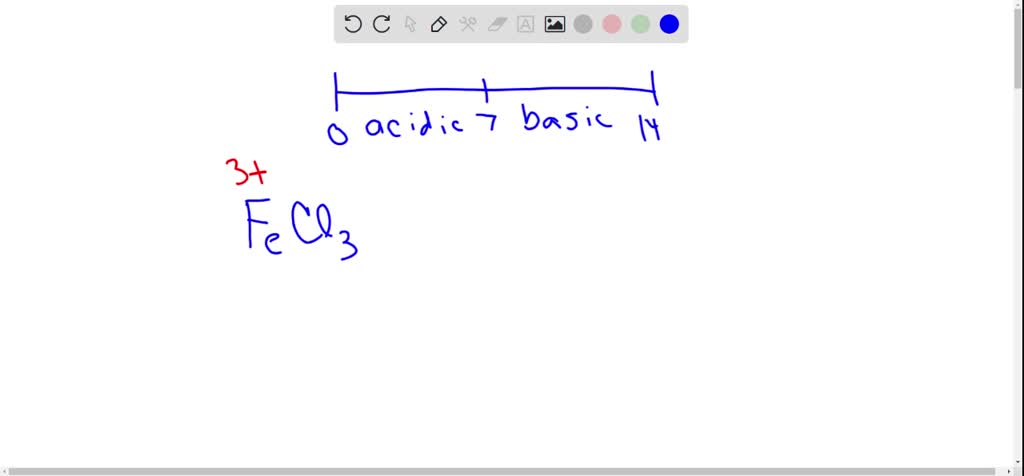
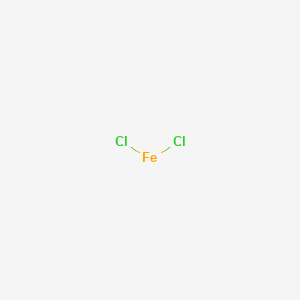
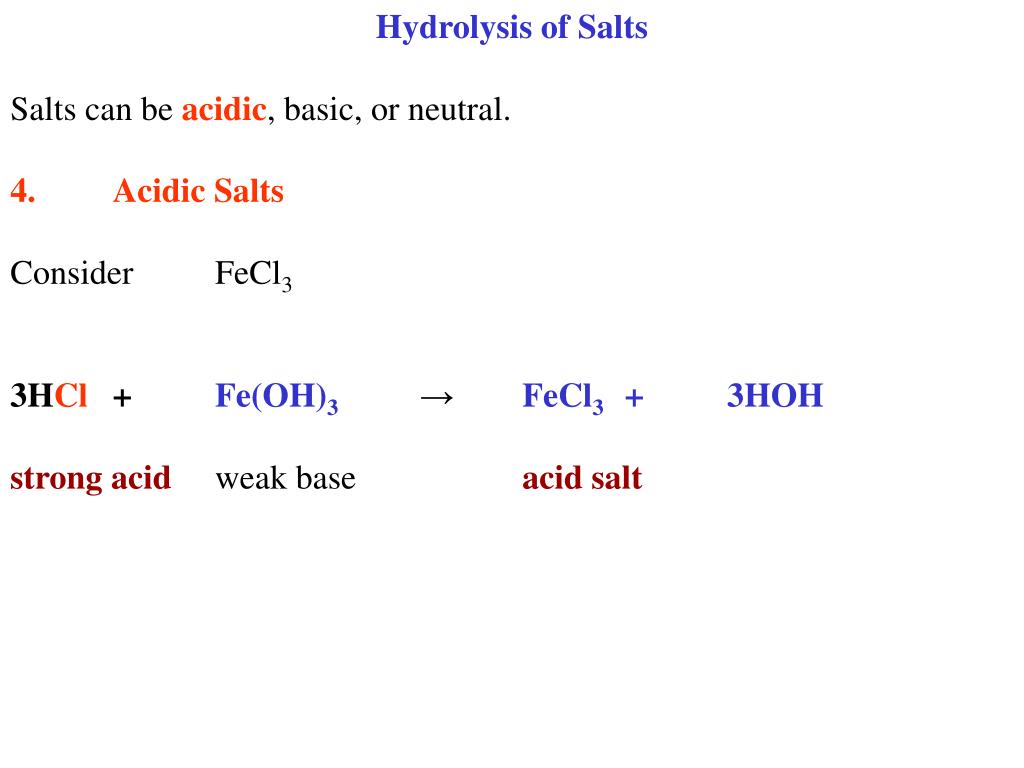
SOLVED: Metallic ions with a higher positive charge are more strongly hydrated and tend to be more acidic in solution. Comparing a 0.12 M FeCl3 solution to a 0.12 M FeCl2 solution,